
UHPLC
UHPLC
 Ultra-High Performance Liquid Chromatography a milestone in the evolution of LC in that columns packed with <2µm particles, used with instrumentation capable of handling the resulting high back pressures, make possible extremely fast and efficient separations. UHPLC is a very powerful tool for today’s practicing chromatographer, as it can significantly increase the efficiency of a chromatographic separation. In addition, the wider range of usable flow rates makes high speed separations possible.
Ultra-High Performance Liquid Chromatography a milestone in the evolution of LC in that columns packed with <2µm particles, used with instrumentation capable of handling the resulting high back pressures, make possible extremely fast and efficient separations. UHPLC is a very powerful tool for today’s practicing chromatographer, as it can significantly increase the efficiency of a chromatographic separation. In addition, the wider range of usable flow rates makes high speed separations possible.
- Instruments
- UHPLC Columns
- UHPLC Performance on any LC instrument
- Applications and support
- Reagents
Effect on efficience
The lower the particle size, the higher column efficience is (see graph below). With particle size decrease the column back pressure increases significantly. This leads to ultra high pressure of longer LC columns. It means that we cannot use 1,9µm UHPLC column the same lenth like 5µm column (eg. 250 mm long column). In the end we have lower or similar column efficieny as we have with standard HPLC columns. The main benefit of the UHPLC columns is the analysis run time, not the efficiency.

If you need to increase efficiency, look at phase chemistry first. In this case look at stationary phase list.
UHPLC Reagents
 The UHPLC technology requires far better quality solvents than what is currently available on the market. Biosolve ULC/MS solvents, buffers and acids line combine the highest demands for:
The UHPLC technology requires far better quality solvents than what is currently available on the market. Biosolve ULC/MS solvents, buffers and acids line combine the highest demands for:
- UV low gradient drift
- Minimal peak impurities
- Lowest ionic background in MS detection
- Less than 100 ppb of Alkali metal
ULC/MS solvents, are micro filtered at 0,1 µm, have a residue after evaporation of max. 1 ppm and are packed under inert gas for better shelf-life. Besides the standard 2,5 l packaging, Biosolve is now offering for nano LC/MS:
- 500 ml bottles of acetonitrile, methanol, and isopropanol
- 1 l bottle of water
- 100 ml TFA
Information about the reagencies are available. Just ask for it.
Applications and support in UHPLC
 Scale down (Method transfer from conventional HPLC to UHPLC)
Scale down (Method transfer from conventional HPLC to UHPLC)
Sample throughput (How do small particles increase the sample throughput?)
Scale down in UHPLC
Scale-down procedure from conventional HPLC to UHPLC requires optimizing columns selectivity and efficiency. As soon as we have finished this method development, we can perform a scale-down procedure. A few simple calculations can be used to determine equivalent run conditions. This article descibes them sequentially.
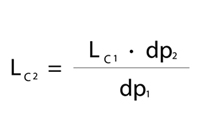 Adjusting Column Size
Adjusting Column Size
The first calculation determines the appropriate column length. Keeping the same column length while decreasing the particle size will increase the number of theoretical plates in that given column length. Therefore, column length can be shortened without losing resolution. By using Equation 1 and when adjusting the column length properly, we can maintain the same separation.
Adjusting Injection Volume
Once we have determined the proper column length, we can determine the appropriate injection volume. Decreasing the column internal diameter and length, decreases the overall column volume and sample capacity. Therefore, we must alter the injection volume as described in Equation 2. Please note that since overall column volume has decreased, it is important to match the sample solvent to the starting mobile phase composition. Mismatched sample solvents can cause irreproducible retention times, efficiencies, and even changes in selectivity.
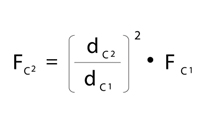 Adjusting Flow Rate
Adjusting Flow Rate
Flow rate must be adjusted to maintain comparable linear velocity through a column with smaller internal diameter. Linear velocity is defined as the distance mobile phase travels over time, whereas flow rate is the volume of mobile phase that travels over time. To maintain the same linear velocity, which is important to maintain efficiencies, flow rates must be decreased as column internal diameter decreases. Also, since smaller particle sizes give rise to higher optimal linear velocities, isocratic flow rates should be calculated with particle size taken into account. Equation 3 can be used to simply and quickly estimate the adjusted flow rate needed for equivalent chromatography. It is also important to note that <2µm particle sizes are less affected by higher flow rates, and therefore faster flow rates can be used in isocratic systems without detrimental effects on peak efficiency.
Adjusting Time Program
Lastly, after we have determined the proper column length, injection volume, and flow rate, we can find the equivalent time needed for gradient or step elutions. As an analytical method is scaled down, the time program needs to also be scaled down to keep the phase interactions the same. Time can be adjusted using Equation 4.
Article referene: Rick Lake, Restek Coroporation





 0
0
 0
0