0
Porovnat
0
Porovnat
 Uživatel
Uživatel
 0
Košík
0
Košík

Rady a tipy
Automatické čištění a kalibrace pH senzorů v procesním měření
V procesním měření pH se často setkáváme s problémem, kdy je potřeba poměrně častá údržba pH senzoru. Při manipulaci se senzorem se bohužel stává, že dojde k jeho poškození. Aby se mu předešlo a aby bylo možné provádět častější údržbu bez zásahu člověka, je možné investovat do technologie a vytvořit tak plně automatický systém (cCare), který bude zajišťovat:
- Automatické měření
- Automatické čištění senzoru bez zásahu člověka (žádné riziko kontaminace)
- Automatickou kalibraci
- Výměnu senzoru bez nutnosti odstavit provoz

Takovýto systém lze sestavit díky inovativnímu řešení firmy Knick. Toto řešeno je založeno na sestavě, kterou tvoří:
- Inteligentní převodník Protos
- Automatická vytahovací armatura Ceramat nebo Sensogate
- Systém Unical 9000
- Senzor Memosens
Systém je možné instalovat přímo v provozu jak v bezpečném prostředí, tak i v prostředí s nebezpečím výbuchu. Armatury mohou být umístěny jednak přímo do potrubí jednak i do reaktoru. Připojení mohou být různá, počínaje standardní přírubou konče speciálními přípojkami Variline.
Měření pH
Výběr pH elektrody
Skleněné × epoxidové tělo
Díky mechanické odolnosti jsou elektrody s tělem z epoxidové pryskyřice vhodné pro "hrubší" zacházení a náročné terénní podmínky, jejich nevýhodou je jejich nižší tepelná odolnost ve srovnání s elektrodami skleněnými. Nyní je lze rovněž použít pro stanovení pH v roztocích obsahujících organická rozpouštědla a korozívní látky. Skleněné elektrody naopak snesou vyšší pracovní teploty a vysoce korozívní rozpouštědla.
Jednomůstková × dvojmůstková
Ekonomické jednomůstkové elektrody jsou vhodné pro většinu obecných aplikací. Dvojmůstkové elektrody, u kterých nehrozí kontaminace referenčního můstku, je vhodné použít ke stanovení pH roztoků obsahujících sulfidy, těžké kovy nebo tris pufry.
Plnitelná × uzavřená (bezúdržbová)
Plnitelné elektrody umožňují díky přítomnosti plnícího otvoru doplnění nebo výměnu elektrolytu referenční elektrody - což je ekonomické a zaručuje dlouhodobou životnost elektrody. Uzavřené elektrody jsou mechanicky velice odolné a nevyžadují údržbu. Mají však obvykle kratší životnost.
Ag / AgCl (argentochloridová) × Hg / Hg2Cl2 (kalomelová)
Nejběžnějším typem pH elektrod,vhodným pro všechny standardní aplikace (teplotní limit 80 °C), jsou elektrody založené na elektrochemickém článku Ag / AgCl. Kalomelová elektroda (článek Hg / Hg2Cl2; teplotní limit 70 °C) je doporučená pro stanovení pH roztoků obsahujících proteiny, organické látky nebo těžké kovy schopné reagovat se stříbrem a zanášet tak kapalinový spoj.
Všeobecně použitelná × speciální aplikace
Mezi analyty, jejichž měření nepovažujeme za standardní aplikace (vhodné pro použití všeobecně použitelných elektrod) lze zařadit: roztoky s obsahem těžkých kovů, proteinů, organických rozpouštědel, s vysokým obsahem sodných iontů, sulfidů, roztoky s nízkým obsahem iontů (deionizovaná voda) a tris pufry.
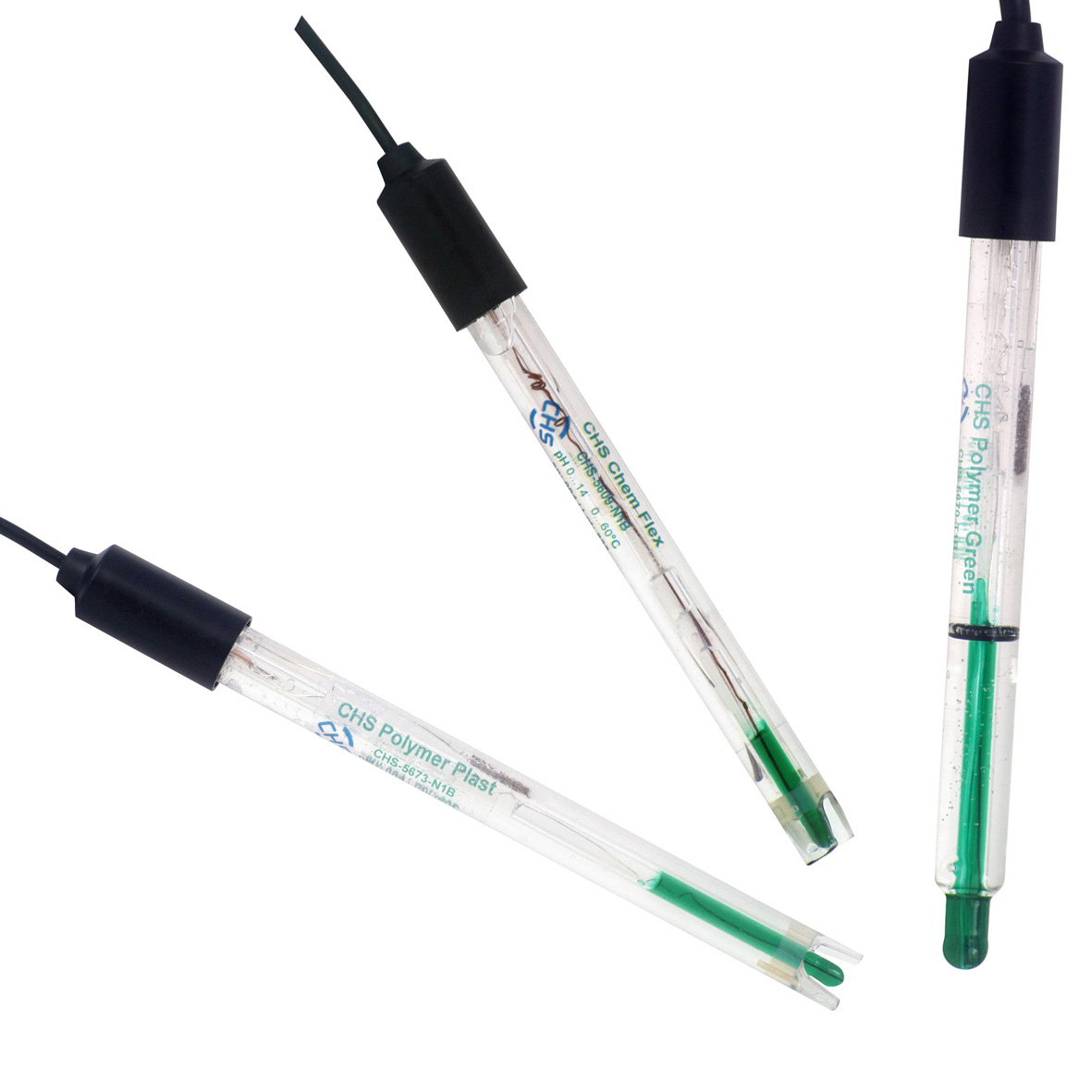
CHS sensors - nová technologie
Laboratorní sondy CHS jsou elektrochemické senzory vyráběné ve Švýcarsku pro měření pH různých médií. Každá sonda má na svém těle název, sériové číslo, číslo šarže a teplotní rozsah. Všechny elektrody podléhají dvojí kontrole kvality, jak ve výrobním závodu, tak před příjmem na sklad Chromservis s.r.o. K sondám je na základě vizuální kontroly a kontroly odezvy certifikovaným pufrem vystavován QC certifikát.
Sondy vyrábíme a nabídku upravujeme podle aktuálních požadavků zákazníků. Dodáváme verze s hlavou S7, s fixním kabelem zakončeným BNC nebo DIN konektorem a verze s teplotním čidlem (NTC 30 kΩ).
DESIGN LABORATORNÍCH ELEKTROD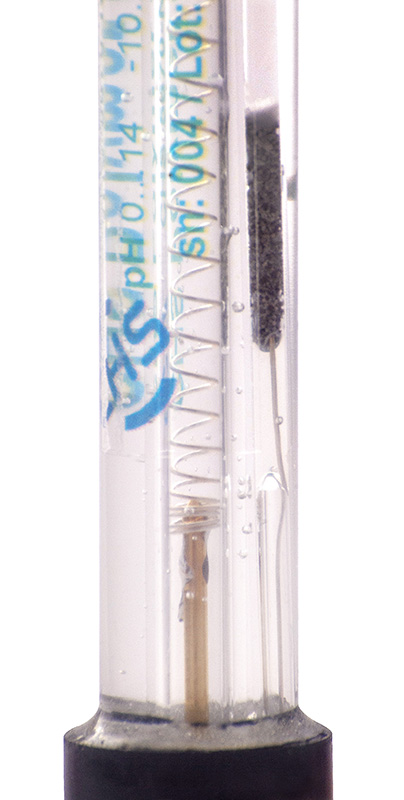
- všechny elektrody mají na těle vytištěný název, sériové číslo, číslo šarže a provozní teplotu
- zelený vnitřní pufr umožňuje vizuální kontrolu pH baňky
- elektrody se dodávají se skladovacím roztokem a uzávěrem
- snadné odstranění krytu vyšroubováním, bezpečné utěsnění krytu
- kvalitní těsnění mezi hlavou elektrody a kabelem splňuje IP68
DESIGN PROCESNÍCH ELEKTROD
- všechny elektrody mají na těle vytištěný název, sériové číslo, číslo šarže a provozní teplotu
- modrý vnitřní pufr zajišťuje snadnou vizuální kontrolu pH baňky
- elektrody jsou dodávány se skladovacím roztokem a uzávěrem
- podle typu jsou elektrody vybaveny teplotním čidlem a tomu odpovídajícím konektorem
Můžete věřit svému pH pufru?
 GMP, GLP, ISO 9001, EN 45000, kalibrace, ověřování, metrologická návaznost a certifikace od akreditované organizace, to jsou stále důležitější pojmy, se kterými se můžete téměř denně setkat v laboratorní praxi. Důležitými „nástroji“ pro měření fyzikálně-chemických veličin jsou i pH a ORP elektrody a právě jejich kalibrace nebyla nikdy tak snadná. Všechny kalibrační postupy předpokládají, že označené hodnoty kalibračního roztoku jsou správné. Ale hodnoty pH pufru se mohou v průběhu času měnit, a tím mohou ovlivnit vaše výsledky měření pH.
GMP, GLP, ISO 9001, EN 45000, kalibrace, ověřování, metrologická návaznost a certifikace od akreditované organizace, to jsou stále důležitější pojmy, se kterými se můžete téměř denně setkat v laboratorní praxi. Důležitými „nástroji“ pro měření fyzikálně-chemických veličin jsou i pH a ORP elektrody a právě jejich kalibrace nebyla nikdy tak snadná. Všechny kalibrační postupy předpokládají, že označené hodnoty kalibračního roztoku jsou správné. Ale hodnoty pH pufru se mohou v průběhu času měnit, a tím mohou ovlivnit vaše výsledky měření pH.
Kompletní řada patentovaných pH pufrů poskytuje stabilitu hodnoty pH, které dříve nebylo dosaženo. Hamilton poskytuje záruku na pH pufry DURACAL po dobu 5 let od data výroby. Roztoky pH 9,21 a pH 10,01 jsou dokonce stabilní na vzduchu. Vysoká pufrovací kapacita umožňuje rychlou a stabilní kalibraci. pH pufry Hamilton DURACAL obsahují konzervační prostředky pro zabránění růstu plísní a mikroorganismů.
Metrologická návaznost
Důležitou otázkou při výrobě certifikovaných referenčních materiálů je zajištění metrologické návaznosti nepřerušeným řetězcem porovnání s referenčním materiálem nejvyšší metrologické kvality (primární referenční materiál).
- Kruhová metrologická návaznost: Na rozdíl od jiných výrobců, kteří používají metrologickou návaznost „směrem dolů“ ("top-down traceability“) Hamilton používá kruhovou metrologickou návaznost, a tedy zpětné sledování návaznosti. Kruhová metrologická návaznost zajišťuje uživatelům pH pufrů DURACAL jedinečnou spolehlivost.
- Metrologická návaznost „nahoru-dolů“: Hamilton stanovuje hodnotu pH pufru DURACAL porovnáním proti dvěma sekundárním referenčním materiálům. Jedná se o referenční materiály od akreditovaných dodavatelů pro sekundární referenční materiály. Takto vyrobené roztoky jsou zpětně porovnávány s primárními referenčními materiály PTB1) nebo NIST2).
- Metrologická návaznost směrem dolů: Aby byla zajištěna nejvyšší možná přesnost a spolehlivost hodnoty pH pufru, je reprezentativní počet vzorků z každé výrobní šarže odeslán německé laboratoři DKD3) (DKD-K-06901) pro externí, nezávislé a nestranné ověření. V této laboratoři jsou vzorky pufrů DURACAL porovnávány se sekundárním referenčním materiálem DKD-K-06901.
Sekundární referenční materiál je samozřejmě navázán na primární referenční materiál od PTB. V této fázi je kruh návaznosti uzavřen: primární referenční materiál PTB je počátečním a koncovým bodem. DKD poskytuje firmě Hamilton kalibrační certifikát pro každý pH pufr DURACAL dané výrobní šarže. Tak je zaručena nejvyšší kvalita pufrů Hamilton, se kterými následně můžete pracovat ve své laboratoři.
Čištění a kondicionování pH elektrod
 pH metry se většinou používají v aplikacích, kdy se vyžaduje pravidelné čištění elektrody. Mezi takové aplikace patří měření vod s vysokou tvrdostí, kalů, vizkozních materiálů nebo vzorků s vysokým obsahem oleje nebo proteinů. Při čištění elektrod dbejte bezpečnostních zásad, jelikož se pracuje s nebezpečnými chemikáliemi: detergenty, HCl (kyselina chlorovodíková) a NaOH (hydroxid sodný).
pH metry se většinou používají v aplikacích, kdy se vyžaduje pravidelné čištění elektrody. Mezi takové aplikace patří měření vod s vysokou tvrdostí, kalů, vizkozních materiálů nebo vzorků s vysokým obsahem oleje nebo proteinů. Při čištění elektrod dbejte bezpečnostních zásad, jelikož se pracuje s nebezpečnými chemikáliemi: detergenty, HCl (kyselina chlorovodíková) a NaOH (hydroxid sodný).
Metoda č.1
Ponořte elektrodu do 0.4 molární HCl (kyselina chlorovodíková) na dobu 10 minut. Potom opláchněte elektrodu deionizovanou nebo destilovanou vodou. Tímto postupem byste měli odstranit organické nečistoty (proteiny) z elektrody a povrchu referenční elektrody.
Metoda č.2
Ponořte elektrodu do 3.8 až 4.0 molárního roztoku KCl (chlorid draselný) vyhřívaného na teplotu 50°C po dobu 1 hodiny. Roztok KCl zchlaďte na laboratorní teplotu a opláchněte elektrodu deionizovanou nebo destilovanou vodou. Tímto postupem zbavíte referenční elektrodu kontaminantů.
Metoda č.3
Ponořte elektrodu do pufru pH=4.0 vyhřívaného na teplotu 50°C po dobu 1 hodiny. Roztok pufru zchlaďte na laboratorní teplotu a opláchněte elektrodu deionizovanou nebo destilovanou vodou. Tímto postupem vyčistíte referenční elektrodu.
Metoda č.4
Po každém použití opláchněte elektrodu 0.5 N nebo 1% roztokem HCl. Pokud se na elektrodě usadily olejové nebo proteinové nečistoty, opláchněte elektrodu teplým roztokem detergentu. Je-li znečištění veliké, ponechejte elektrodu v roztoku detergentu přes noc. Potom opláchněte elektrodu deionizovanou nebo destilovanou vodou a ponořte ji na 10 minut do 1% HCl. Opláchněte elektrodu deionizovanou nebo destilovanou vodou a otestujte ji pomocí pH pufrů. Pokud jste schopni elektrodu správně nakalibrovat, můžete jí dále používat. V opačném případě zopakujte čištění nebo vyměňte elektrodu.
Metoda č.5
Proteiny odstraňte z elektrody takto: ponořte elektrodu do enzymatického roztoku určeného k čištění kontaktních čoček a ponechejte v něm elektrodu přes noc. Enzym zbaví elektrodu proteinů.
Údržba pH elektrod
Údržba a skladování pH elektrod
pH elektrody jsou citlivé senzory, které vyžadují správné zacházení a údržbu, aby poskytovaly přesné a spolehlivé výsledky a měly co nejdelší životnost. Není-li pH elektroda používána delší dobu, je potřeba ji uchovávat ve vlhkém stavu. pH elektrody uchovávejte ve skladovacím roztoku ("storage solution") nebo v pufru pH 7.
NESKLADUJTE elektrodu v destilované nebo deionizované vodě, protože by došlo ke ztrátě iontů ze skleněné baňky elektrody a z referenčního elektrolytu. To by mělo za následek pomalou odezvu.
pH elektrody bývají dodávány buď s ochranným krytem nebo v skladovacích lahvičkách, aby se předešlo zlomení nepo poškrábání elektrody. Navíc je takto pH elektroda uložena ve vlhkém stavu. Před použitím pH elektrodu opatrně vyjměte (nebo sundejte ochranný kryt) a opláchněte ji destilovanou vodou. Při dlouhodobém skladování uchovávejte elektrodu v skladovací lahvičce tak, aby byla skleněná baňka (pH citlivé sklo) dostatečně ponořena. Podle poptřeby vždy doplňujte uchovávací roztok.