

 0
Porovnat
0
Porovnat
 Uživatel
Uživatel
 0
Košík
0
Košík

Měřící principy
Katalytické spalování
Princip
 Pellistor se skládá ze dvou spirálek tenkého platinového drátku zalitých v hliníkových perličkách a zapojených do Wheatsonova můstku. Jedna z perliček je impregnována speciálním katalyzátorem podněcujícím oxidaci hořlavých plynů (par), naopak druhá je upravená pro inhibici oxidace. Platinovými spirálkami prochází elektrický proud a zahřívají se na teplotu, při které dojde k oxidaci přítomných hořlavých plynů (par) na katalyzátoru. Oxidační proces dále zvyšuje teplotu hliníkové perličky s katalyzátorem, zahřívá platinovou spirálku a tím zvyšuje její elektrický odpor. To má za následek elektrickou nerovnováhu můstkového zapojení.
Pellistor se skládá ze dvou spirálek tenkého platinového drátku zalitých v hliníkových perličkách a zapojených do Wheatsonova můstku. Jedna z perliček je impregnována speciálním katalyzátorem podněcujícím oxidaci hořlavých plynů (par), naopak druhá je upravená pro inhibici oxidace. Platinovými spirálkami prochází elektrický proud a zahřívají se na teplotu, při které dojde k oxidaci přítomných hořlavých plynů (par) na katalyzátoru. Oxidační proces dále zvyšuje teplotu hliníkové perličky s katalyzátorem, zahřívá platinovou spirálku a tím zvyšuje její elektrický odpor. To má za následek elektrickou nerovnováhu můstkového zapojení.
Výhody
- Lineární závislost až do 100% DMV
- Levný a stabilní senzor
- Vysoká rychlost odezvy (< 10 s)
- Rozsah pracovní teploty -40 až +60°C
Nevýhody
- Náchylnost na otravu katalyzátoru a tím snížení citlivosti
- Pro svou činnost vyžaduje atmosféru s obsahem minimálně 10% kyslíku
- „Otrávený“ pellistor dává signál jako při nulové koncentraci, proto je nutno ho ověřovat kalibračním plynem
- Vyšší energetická náročnost
Elektrochemický senzor
Princip
Elektrochemický článek je systém 2, 3 popř. 4 elektrod, které jsou umístěny v gelovém elektrolytu. Prostor s elektrolytem a elektrodami je oddělen od okolní atmosféry difúzní bariérou. Tou procházejí molekuly měřeného plynu, které reagují s elektrolytem. Na elektrodách dochází k oxidační a redukční reakci, která má za následek změnu potenciálu článku. Se vzrůstající koncentrací plynu vzrůstá i potenciál.
Výhody
- Pro „běžné“ plyny se jedná o spolehlivé a levné články
Nevýhody
- Dlouhá odezva (v některých případech se jedná i o minuty)
- Vysoká cena pro speciální plyny
- Možnost poškození vysokou koncentrací plynu
- Křížové interference (u senzoru ozonu se jedná i o vliv prouděním vzduchu, teploty a vlhkosti)
Fotoionizační detektor
Princip
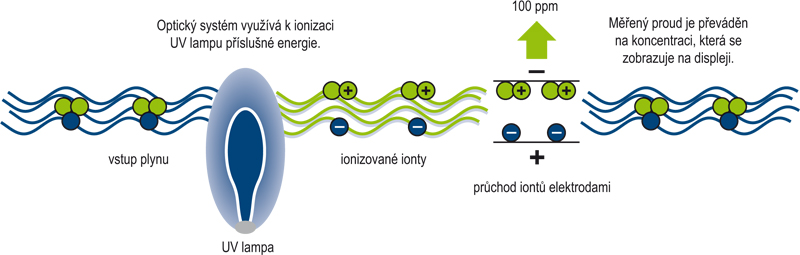
Fotoionizační detektor (Photo Ionization Detector) pracuje na principu měření elektrického náboje vzniklého při ionizaci měřeného plynu. U většiny plynů lze určit tzv. specifický ionizační potenciál (IP), který má jednotku eV. Měřený plyn je ionizován ultrafialovou zářivkou, což se projeví vznikem elektrického náboje. Ionizace plynu je však podmíněna skutečností, že ionizační potenciál plynu bude menší než hodnota potenciálu (eV) použité UV lampy (respektive energie vzniklých fotonů)! Vlastní senzor detekuje vzniklý náboj ionizovaného plynu a ten je převeden na elektrický proud. Proud je zesílem a převeden na koncentraci v jednotkách ppm nebo ppb.
Výhody
- moderní typ (3D) nepodléhá vlivům teploty ani vlhkosti
- jedním detektorem lze měřit široké spektrum látek
- vysoká citlivost (jednotky ppb)
- vynikající rychlost odezvy (< 3 s)
- vysoká přesnost i při velice nízkých koncetracích
Nevýhody
- pro většinu sloučenin nízká selektivita
Infračervený senzor
Princip
Infračervený detektor (IR) využívá schopnosti plynů se dvěma nebo více atomy (např. oxid uhličitý, metan) absorbovat infračervené světlo (IR).Plyn je v infračerveném detektoru detekován měřením absorpce na určité frekvenci IR záření, která odpovídá vibraci nebo rotaci molekulární vazby mezi rozdílnými atomy. S nárůstem koncentrace měřeného plynu se snižuje úroveň výstupního signálu z IR senzoru (přibližně logaritmická závislost).
Výhody
- detektory měří i v atmosféře bez přítomnosti kyslíku
- nejsou poškozovány katalytickými jedy
- varování při znečištění optiky
- kvalitní detektory pracují až do 80% znečištění optiky
- dobrá selektivita
Nevýhody
- vyšší cena
Tepelně vodivostní detektor (TCD)
 Princip
Princip
Senzor pracuje na principu porovnání tepelné vodivosti vzorku s referenčním plynem (obvykle vzduch). Vyhřívaný termistor nebo platinové vlákno je vystaveno působení měřeného plynu, druhý identický měrný prvek je uzavřen v komůrce s referenčním plynem. Pokud je tepelná vodivost měřeného plynu vyšší než referenčního plynu, teplota měrného prvku se sníží (a naopak). Změna teploty má za následek změnu elektrického odporu a je měřitelná podobně jako u pellistoru.
Výhody
- Vhodný pro binární směsi
- Vysoká citlivost u plynů s vyšší tepelnou vodivostí (He, H2, Ne, CH4)
- Rychlá odezva
Nevýhody
- Výstupní signál značně ovlivňuje změna okolní teploty
- Nepoužitelný pro vícesložkové směsi plynů
- Přítomné plyny s nižší tepelnou vodivostí než vzduch způsobují interference
- Plyny s relativní tepelnou vodivostí blízkou 1 jsou neměřitelné (CO, O2, N2, NH3)




